In the FELIX trial, patients' T-cells were engineered with precision1

*7/153 enrolled patients did not receive AUCATZYL infusion due to manufacturing-related issues.2
†In the pivotal trial, the median time from leukapheresis to product release was 21 days (range, 18 to 50).2
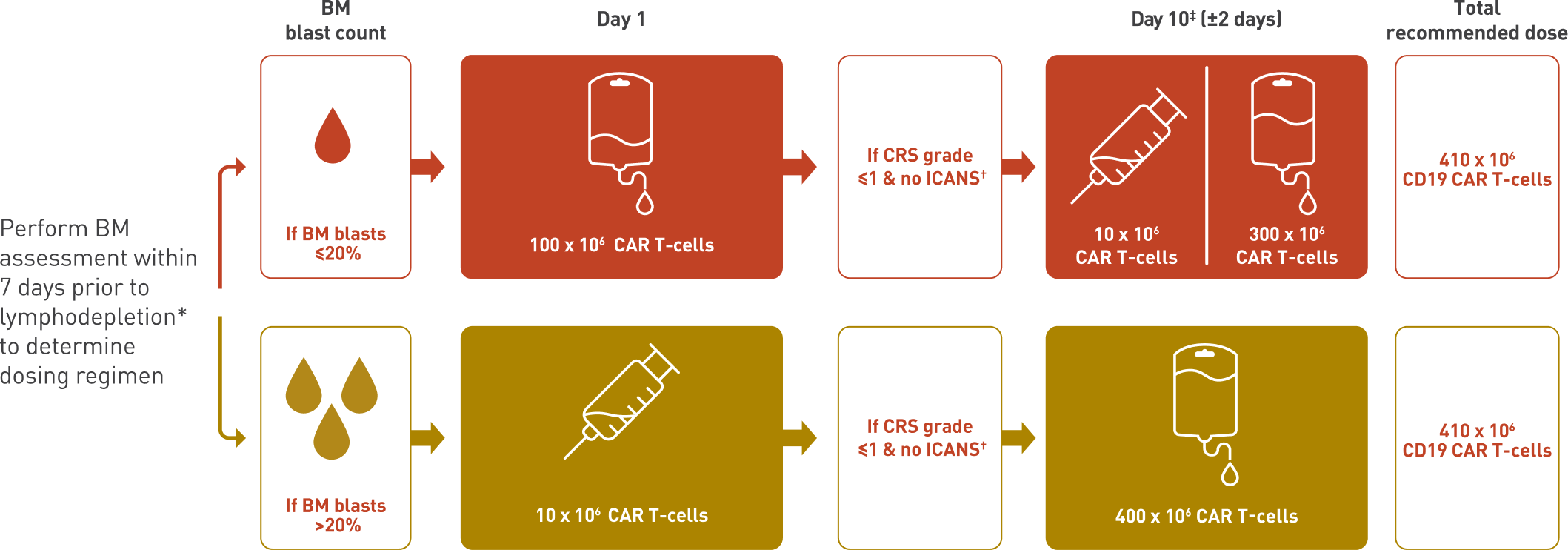
AUCATZYL is administered based on your patients’ tumour volume1
Because higher disease burden increases the risk of CRS and ICANS, AUCATZYL is dosed according to tumour volume:1,3
Patients with BM blast count ≤20% start with a higher first dose
Patients with BM blast count >20% start with a lower first dose
Customised split dosing on Days 1 & 10 (±2 days)1
AUCATZYL is for autologous and IV use only.
Treatment centres must have 24-hour immediate access to tocilizumab and emergency equipment must be available prior to infusion.
Refer to the UK Public Assessment Report on the MHRA website for details of leukapheresis, lymphodepletion and bridging therapies used in the clinical trials for AUCATZYL.
*Administer the lymphodepleting chemotherapy regimen before infusion of AUCATZYL: FLU 30 mg/m2/day IV and CY 500 mg/m2/day IV. FLU and CY will be given together for 2 days and FLU alone on the third and fourth day (total dose: FLU 120 mg/m2; CY 1000 mg/m2).1,2
†If patient has Grade 2 CRS, consider postponing AUCATZYL infusion up to Day 21 to allow for CRS to resolve to Grade ≤1. If patient has Grade 1 ICANS, consider postponing AUCATZYL infusion up to Day 21 to allow for the ICANS to completely resolve.1
‡The second dose can be avoided or delayed if there are signs of severe immune toxicity.1
AUCATZYL treatment schedule1
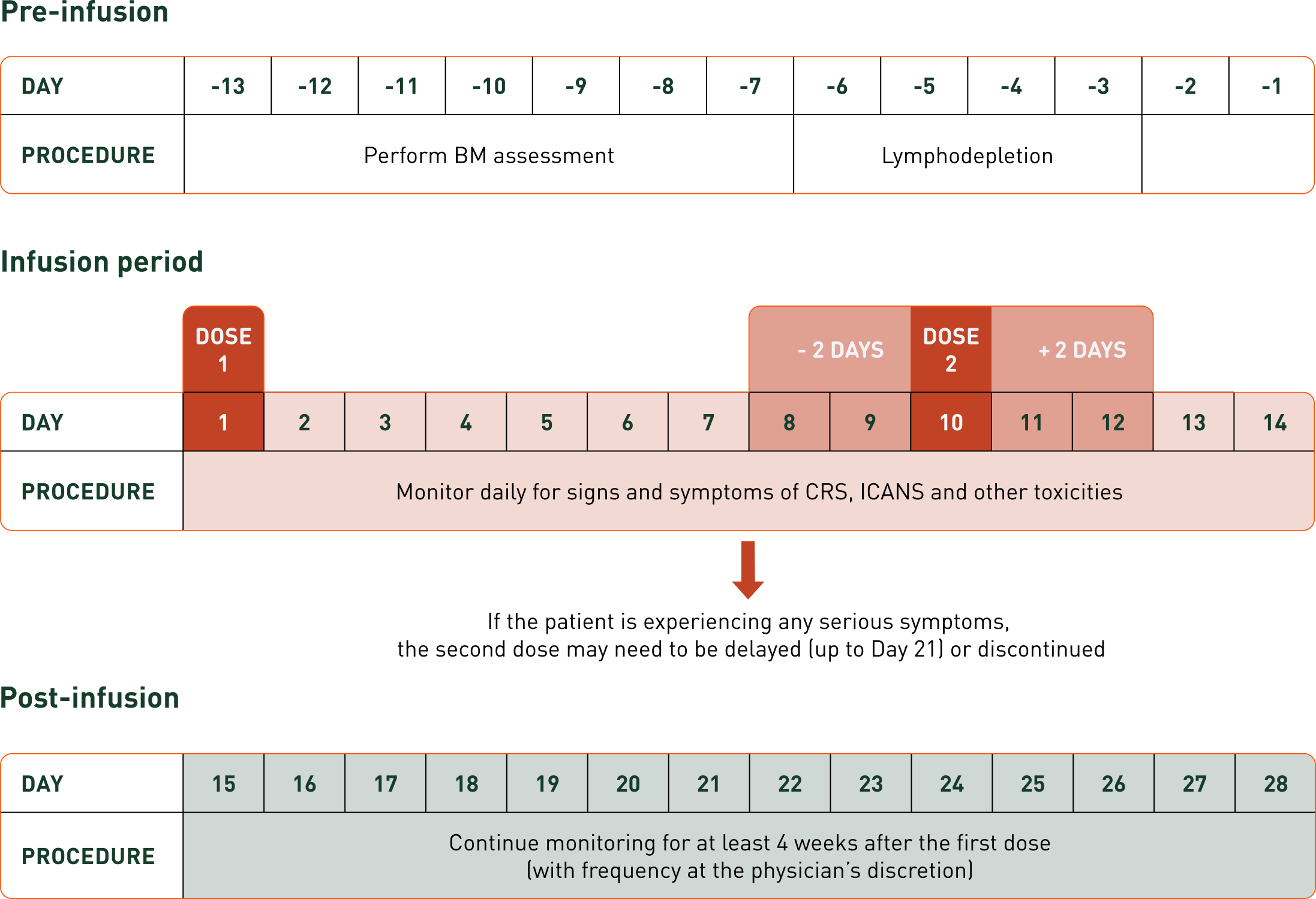
This specific lymphodepleting regimen (chosen drugs and days dosed) was administered in the FELIX trial. Healthcare professionals may select a preferred alternative lymphodepletion regimen in actual practice.
Treatment centres must have 24-hour immediate access to tocilizumab and emergency equipment must be available prior to infusion.
Please refer to the summary of product characteristics for special precautions for storage, disposal and other handling.


Stay updated
Sign up to receive the latest news and product updates from Autolus.
By clicking ‘Receive key updates’ you’re confirming that you agree with our Terms and conditions.
Aucatzyl is indicated for the treatment of adult patients (≥18 years) with relapsed or refractory B-cell precursor acute lymphoblastic leukaemia.1
NICE recommends AUCATZYL to treat relapsed or refractory B-cell precursor acute lymphoblastic leukaemia in people aged ≥26 years in England, Wales and Northern Ireland.4,5
BM=bone marrow, CAR=chimeric antigen receptor, CRS=cytokine release syndrome, CY=cyclophosphamide, FLU=fludarabine, ICANS=immune effector cell-associated neurotoxicity syndrome, IV=intravenously, MHRA=Medicines and Healthcare products Regulatory Agency, NICE=National Institute for Health and Care Excellence.
Summary of Product Characteristics, Aucatzyl 2025.
Roddie C, et al. Obecabtagene autoleucel in adults with B-cell acute lymphoblastic leukemia. N Engl J Med. 2024;391(23):2219–2230.
Turtle C, et al. CD19 CAR–T cells of defined CD4+:CD8+ composition in adult B cell ALL patients. J Clin Invest. 2016;126(6):2123–2138.
NICE. Obecabtagene autoleucel for treating relapsed or refractory B-cell precursor acute lymphoblastic leukaemia. Issued December 2025. Accessed March 2026. https://www.nice.org.uk/guidance/ta1116/resources/obecabtagene-autoleucel-for-treating-relapsed-or-refractory-bcell-precursor-acute-lymphoblastic-leukaemia-pdf-2973530453903557.
AWTTC. Obecabtagene autoleucel (Aucatzyl®). Issued November 2025. Accessed March 2026. https://awttc.nhs.wales/accessing-medicines/medicine-recommendations/obecabtagene-autoleucel-aucatzyl/.


