Patients with relapsed/refractory B-cell precursor acute lymphoblastic leukaemia often have treatment-resistant disease and varied clinical profiles3-5
Despite recent advances, the treatment landscape still does not offer options for all adult patients R/R B-ALL6,7
Allogeneic stemcell transplantation (allo-SCT)
Allo-SCT is the only known curative therapy for relapsed ALL-SCT6
Not always feasible because of patient fitness, donor availability and the ability to achieve a remission prior to an SCT8
Not recommended for patients who have previously received an SCT6
Potential for non-relapse mortality following an allo-SCT, most often caused by infections, toxicities and GVHD9,10
Blinatumomab (CD19 bispecific) and inotuzumab ozogamicin (CD22 ADC)
Demonstrated survival benefit over standard chemotherapeutic regimens11,12
Short-term remission with median OS <1 year11,12
Blinatumomab requires continuous IV infusion13
Inotuzumab ozogamicin is contraindicated for patients with prior liver disease due to risk of sinusoidal obstruction syndrome12,14
Blinatumomab is approved for treatment of R/R B-ALL but is being increasingly used as a first-line treatment15,16
Brexucabtagene autoleucel and tisagenlecleucel (anti-CD19 CAR T-cell therapy)
Revolutionised the treatment landscape by facilitating deep CRs and improving OS over standard-of-care systemic options11,12,17,18
Potential for significant immune-mediated toxicity, with a high frequency of Grade ≥3 adverse events in clinical trials19,20
May require subsequent allo-SCT for long-term benefit21
Allogeneic stemcell transplantation (allo-SCT)
Blinatumomab (CD19 bispecific) and inotuzumab ozogamicin (CD22 ADC)
Brexucabtagene autoleucel and tisagenlecleucel (anti-CD19 CAR T-cell therapy)
Allo-SCT is the only known curative therapy for relapsed ALL-SCT6
Demonstrated survival benefit over standard chemotherapeutic regimens11,12
Revolutionised the treatment landscape by facilitating deep CRs and improving OS over standard-of-care systemic options11,12,17,18
Not always feasible because of patient fitness, donor availability and the ability to achieve a remission prior to an SCT8
Not recommended for patients who have previously received an SCT6
Potential for non-relapse mortality following an allo-SCT, most often caused by infections, toxicities and GVHD9,10
Short-term remission with median OS <1 year11,12
Blinatumomab requires continuous IV infusion13
Inotuzumab ozogamicin is contraindicated for patients with prior liver disease due to risk of sinusoidal obstruction syndrome12,14
Blinatumomab is approved for treatment of R/R B-ALL but is being increasingly used as a first-line treatment15,16
Potential for significant immune-mediated toxicity, with a high frequency of Grade ≥3 adverse events in clinical trials19,20
May require subsequent allo-SCT for long-term benefit21
AUCATZYL: Designed to deliver potency and persistence with next-generation CAR-T construction1,22
Exploring the role of CAT19 and 4-1BB
Dr Eleni Tholouli explains how the unique features of AUCATZYL may support long-term remission and survival by combining persistent efficacy with safety benefits23
Preclinical evaluation of CAT19 binding technology shows potential safety benefits vs FMC63 CAR T-cells:2,22
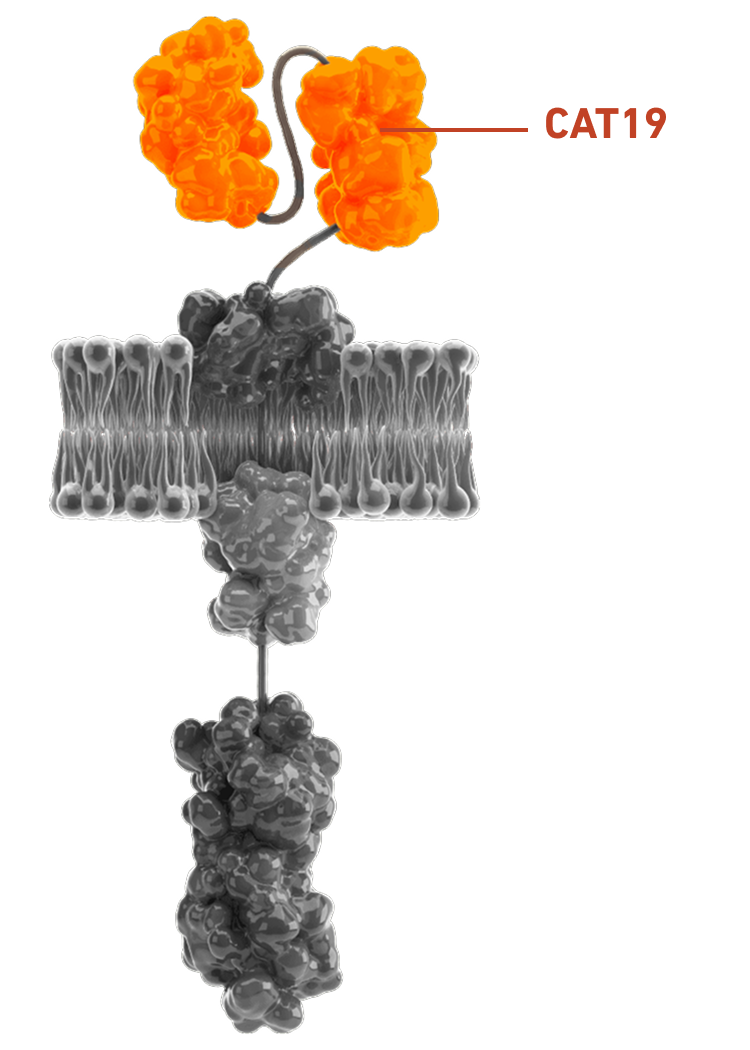
AUCATZYL includes CAT19, a fast off-rate CD19 binder that may help reduce overstimulation while still enabling repeated binding and tumour cell targeting2,22
AUCATZYL's fast off-rate has the potential to reduce the risk of toxicities vs FMC63 CAR T-cells2,22
Slow off-rate: Stays bound longer and may overstimulate T-cells2,22
Fast off-rate: Disengages quickly, which may reduce the risk of overstimulation2,22
Overstimulation can trigger CRS and ICANS24
Preclinical studies show 4-1BB costimulation provides greater potency and persistence vs CD28-based CARs:22,25
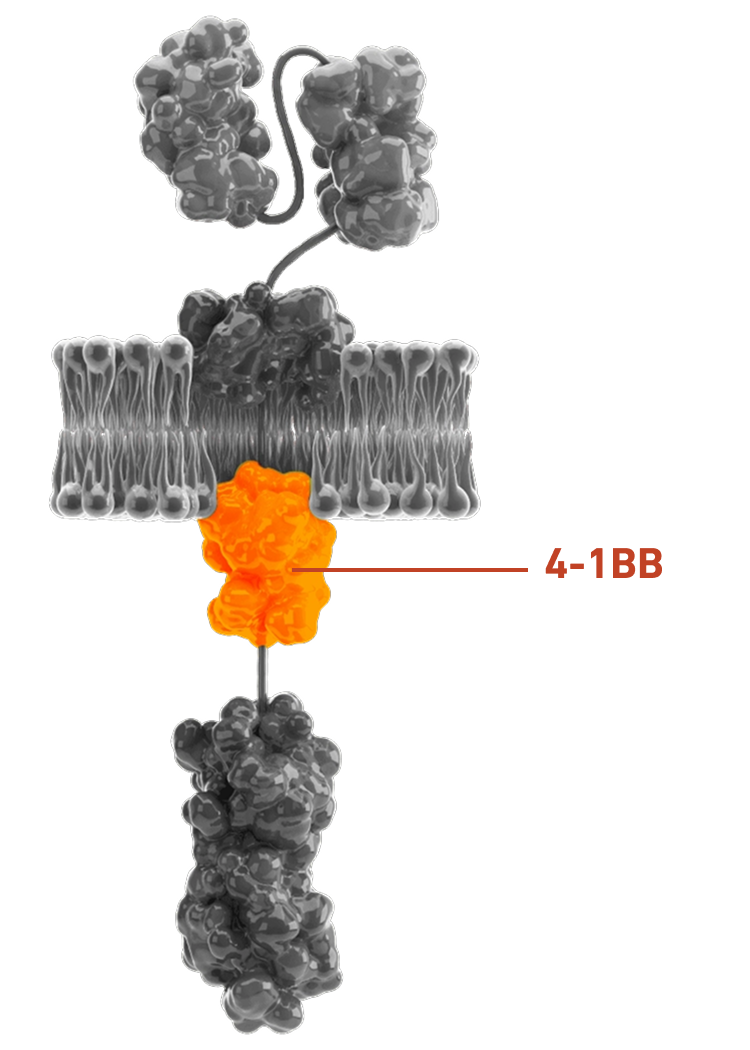
AUCATZYL includes a 4-1BB costimulatory domain, which may improve persistence and help patients maintain a response for longer1,22,25
AUCATZYL’s 4-1BB costimulatory domain may increase potency and persistency vs CD28-based CARs1,25
Enhanced expansion: CAR T-cells multiply more effectively after activation22
Reduced exhaustion: CAR T-cells remain functional for longer22

Clinical data
Explore the efficacy and safety profile of AUCATZYL from the pivotal FELIX trial.

Stay updated
Sign up to receive the latest news and product updates from Autolus.
By clicking ‘Receive key updates’ you’re confirming that you agree with our Terms and conditions.
Aucatzyl is indicated for the treatment of adult patients (≥18 years) with relapsed or refractory B-cell precursor acute lymphoblastic leukaemia.1
NICE recommends AUCATZYL to treat relapsed or refractory B-cell precursor acute lymphoblastic leukaemia in people aged ≥26 years in England, Wales and Northern Ireland.26,27
ADC=antibody-drug conjugate, allo-SCT=allogeneic stem cell transplant, B-ALL=B-cell precursor acute lymphoblastic leukaemia, CAR=chimeric antigen receptor, CR=complete response, CRS=cytokine release syndrome, GVHD=graft-versus-host disease; ICANS=immune effector cell-associated neurotoxicity syndrome, NICE=National Institute for Health and Care Excellence, OS=overall survival, R/R=relapsed/refractory, SCT=stem cell transplant.
Summary of Product Characteristics, Aucatzyl 2025.
Roddie C, et al. Durable responses and low toxicity after fast off-rate CD19 chimeric antigen receptor-T therapy in adults with relapsed or refractory B-cell acute lymphoblastic leukemia. J Clin Oncol. 2021;39(30):3352–3363.
Kayser S, et al. Outcome of relapsed or refractory acute B-lymphoblastic leukemia patients and BCR-ABL-positive blast cell crisis of B-lymphoid lineage with extramedullary disease receiving inotuzumab ozogamicin. Haematologica. 2022;107(9):2064–2071.
Guru Murthy GS and Atallah E. Relapsed/Refractory Acute Lymphoblastic Leukemia in Adults: Progress and Challenges. JCO Oncol Pract. 2022;18(7):489–490.
Lamble AJ, et al. How I treat postimmunotherapy relapsed B-ALL. Blood. 2025;145(1):64–74.
Pan London Haemato RM Partners, South East London Cancer Alliance Network, North Central and East London Cancer Alliance 2020. Oncology Clinical Guidelines: Acute Leukaemias and Myeloid Neoplasms Part 1: Acute Lymphoblastic Leukaemia. Accessed March 2026. https://rmpartners.nhs.uk/wp-content/uploads/2020/01/Pan-London-ALL-Guidelines-Jan-2020.pdf.
Hoelzer D, et al. ESMO Clinical Practice Guideline interim update on the use of targeted therapy in acute lymphoblastic leukaemia. Ann Oncol. 2024;35(1):15–28.
Maffini E, et al. Treatment of Adult Patients with Relapsed/Refractory B-Cell Philadelphia-Negative Acute Lymphoblastic Leukemia. Clin Hematol Int. 2019;1(2):85–93.
Akhmedov M, Espinoza JL. Addressing the surge of infections by multidrug-resistant Enterobacterales in hematopoietic cell transplantation. Blood Rev. 2024;68:101229.
Averbuch D, et al. Antimicrobial resistance in gram-negative rods causing bacteremia in hematopoietic stem cell transplant recipients: intercontinental prospective study of the Infectious Diseases Working Party of the European Bone Marrow Transplantation Group. Clin Infect Dis. 2017;65(11):1819-1828.
Kantarjian H, et al. Blinatumomab for Acute Lymphoblastic Leukemia. N Engl J Med. 2017;376(23):e49.
Kantarjian HM, et al. Inotuzumab ozogamicin versus standard of care in relapsed or refractory acute lymphoblastic leukemia: Final report and long-term survival follow-up from the randomized, phase 3 INO-VATE study. Cancer. 2019;125(14):2474–2487.
Prescribing Information, Blincyto 2024.
Summary of Product Characteristics, Besponsa 2025.
Jabbour EJ, et al. Frontline Ph‑negative B‑cell precursor acute lymphoblastic leukemia treatment and the emerging role of blinatumomab. Blood Cancer J. 2024;14(1):203.
Wilke AC and Gökbuget N. The role of blinatumomab in adult acute B lymphoblastic leukaemia. Br J Haematol. 2025;207(1):27‑42.
Shah BD, et al. Two‑year follow‑up of KTE‑X19, an anti‑CD19 chimeric antigen receptor (CAR) T‑cell therapy, in adult patients with relapsed/refractory B‑cell acute lymphoblastic leukaemia (ZUMA‑3). J Clin Oncol. 2022;40(16_suppl):7010.
Rampotas A and Roddie C. The present and future of CAR T‑cell therapy for adult B‑cell acute lymphoblastic leukaemia. Blood. 2025;145(14):1485‑1497.
Bouchkouj N, et al. FDA Approval Summary: Brexucabtagene Autoleucel for Treatment of Adults With Relapsed or Refractory B-Cell Precursor Acute Lymphoblastic Leukemia. Oncologist. 2022;27(10):892–899.
Frey NV, et al. Optimizing Chimeric Antigen Receptor T-Cell Therapy for Adults With Acute Lymphoblastic Leukemia. J Clin Oncol. 2020;38:415–422.
Hay KA, et al. Factors associated with durable EFS in adult B-cell ALL patients achieving MRD-negative CR after CD19 CAR T-cell therapy. Blood. 2019;133(15):1652–1663.
Ghorashian S, et al. Enhanced CAR T cell expansion and prolonged persistence in pediatric patients with ALL treated with a low-affinity CD19 CAR. Nat Med. 2019;25:1408–1414.
Park JH, et al. Can CAR T-cell therapy be a definitive treatment for adult R/R B-ALL without transplant? Long-term findings and predictors of sustained remission for obe-cabtagene autoleucel. Presentation number: S113. European Hematology Association 2025 Congress. 12–15 June, 2025. Milan, Italy.
Morris EC, et al. Cytokine release syndrome and associated neurotoxicity in cancer immunotherapy. Nat Rev Immunol. 2021;22(2):85–96.
Zhao X, et al. Efficacy and safety of CD28- or 4-1BB-based CD19 CAR-T cells in B cell acute lymphoblastic leukemia. Mol Ther Oncolytics. 2020;18:272–281.
NICE. Obecabtagene autoleucel for treating relapsed or refractory B-cell precursor acute lymphoblastic leukaemia. Issued December 2025. Accessed March 2026. https://www.nice.org.uk/guidance/ta1116/resources/obecabtagene-autoleucel-for-treating-relapsed-or-refractory-bcell-precursor-acute-lymphoblastic-leukaemia-pdf-297353045390557.
AWTTIC. Obecabtagene autoleucel (Aucatzyl®). Issued November 2025. Accessed March 2026. https://awttc.nhs.wales/accessing-medicines/medicine-recommendations/obecabtagene-autoleucel-aucatzyl/.


