The FELIX trial is the largest pivotal CAR T-cell study in adults with R/R B-ALL2,3
Explore the study design, efficacy outcomes and safety profile and hear expert perspectives on how the FELIX findings may help inform clinical practice.
Bringing the data to life
Watch Dr Claire Roddie explore key insights from the FELIX trial and discuss their relevance for clinical practice in eligible adults with R/R B-ALL
FELIX: An open-label, multicentre, multinational, single-arm, phase Ib/II study of AUCATZYL1,2,4
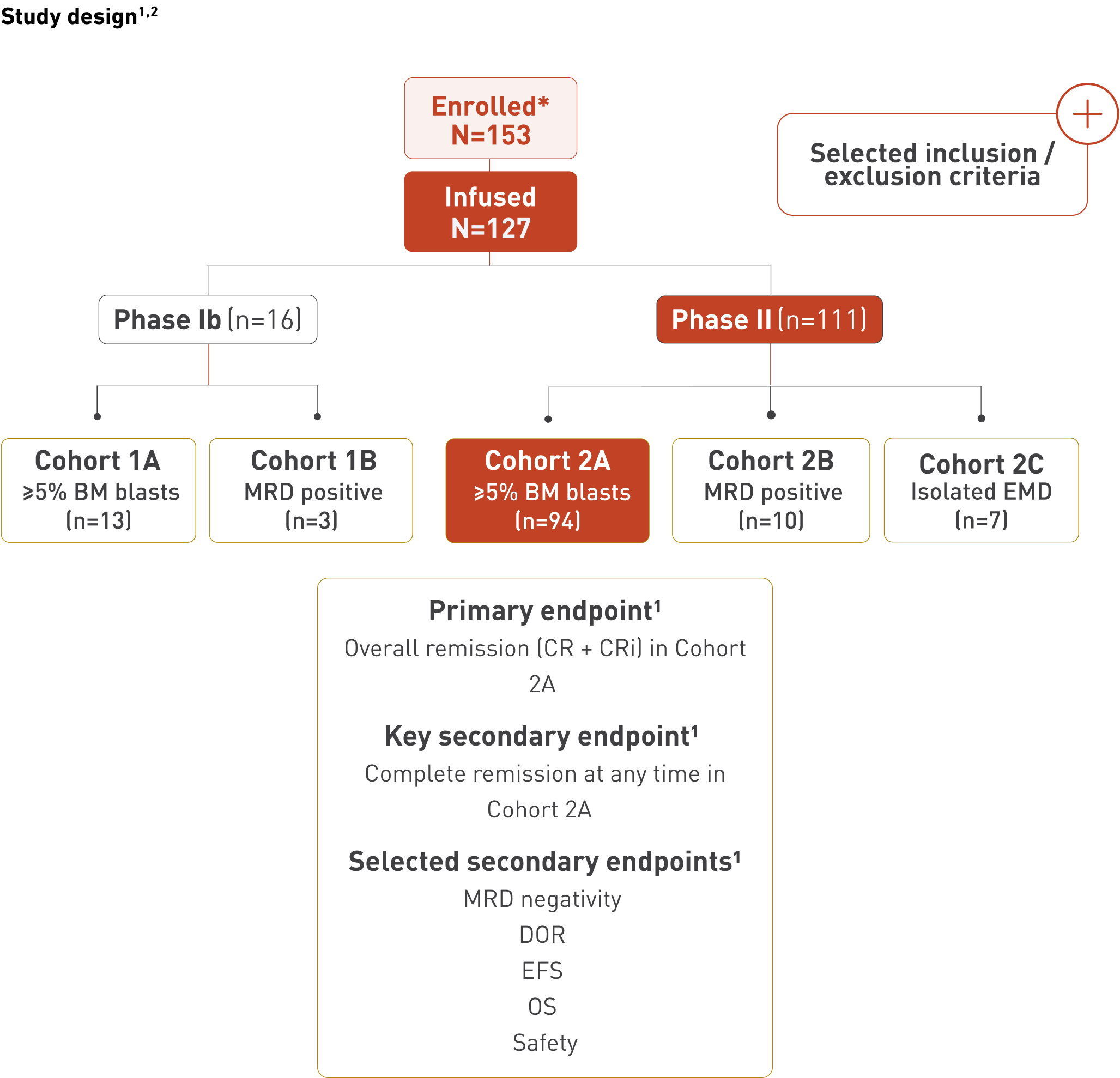
*All eligibility criteria met and leukapheresate accepted for manufacturing. Twenty-six patients discontinued prior to infusion due to: death (15), manufacturing related (7), adverse event (2), physician decision (1), and progressive disease (1).1
The safety data are based on the total infused population (N=127), but the efficacy data for the primary and key secondary endpoints are based on Cohort 2A (n=94) specifically.1,2
AUCATZYL is for autologous and intravenous use only.2
Treatment consisted of lymphodepleting chemotherapy followed by AUCATZYL as a split-dose infusion (Day 1 and Day 10) with a total dose of 410 × 106 CD19 CAR-positive viable T-cells.1,2
Refer to the UK Public Assessment Report on the MHRA website for details on leukapheresis, lymphodepletion and bridging therapies used in the clinical trials for AUCATZYL.
Cohort 2A has been specifically highlighted because it was the pivotal efficacy cohort in which the primary and key secondary endpoints were formally tested, while analysis of the total infused population provided supportive context across all treated patients.1
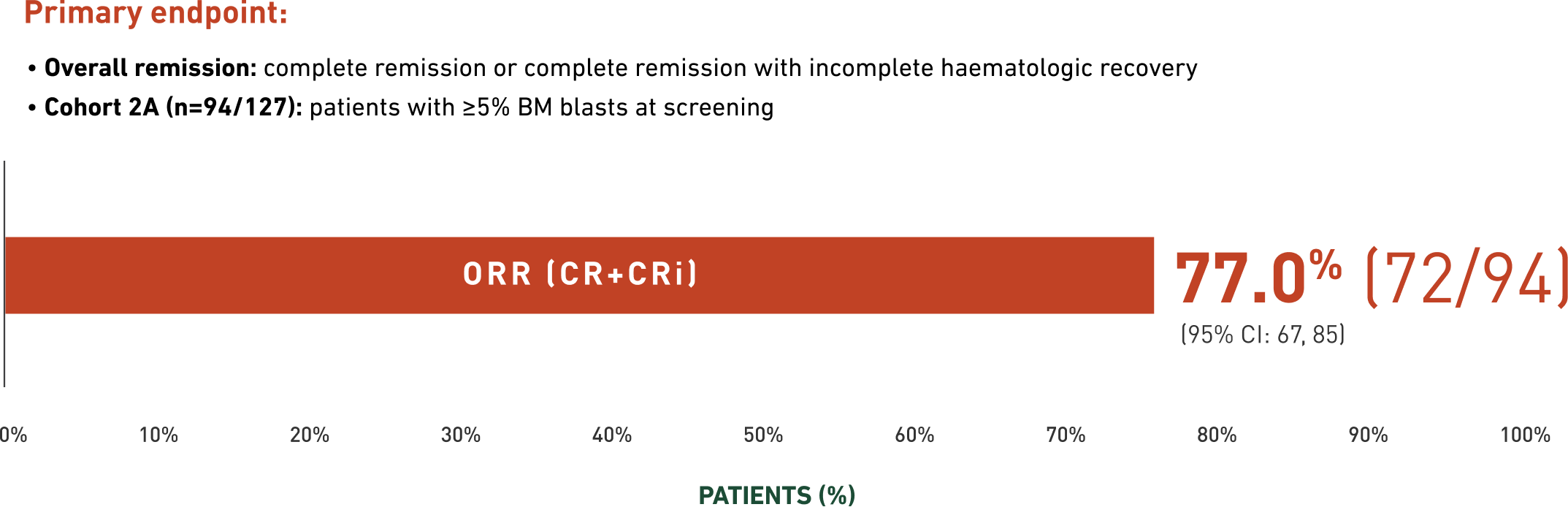
Overall remission in Cohort 2A1
Data cut-off: 7 February 2024; median follow-up: 20.3 months1
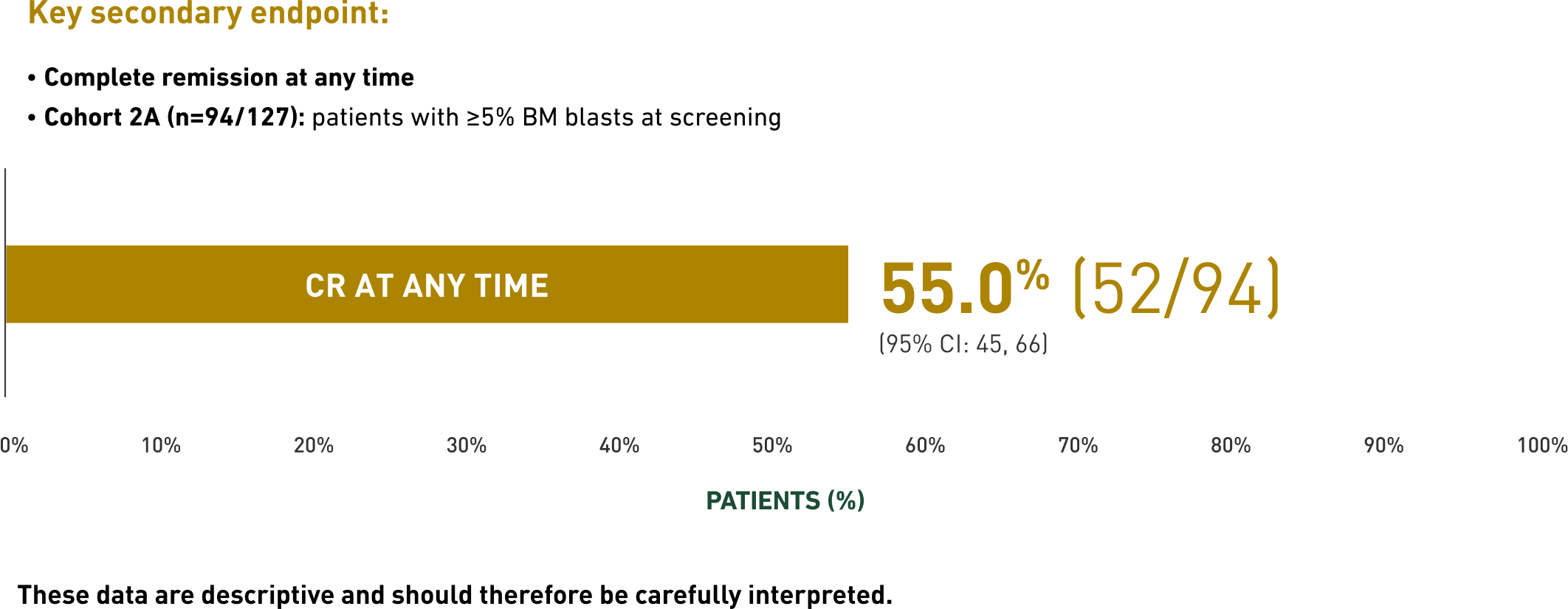
CR at any time1
Selected secondary endpoints
Click on the buttons below to review selected secondary endpoint Cohort 2A data on:
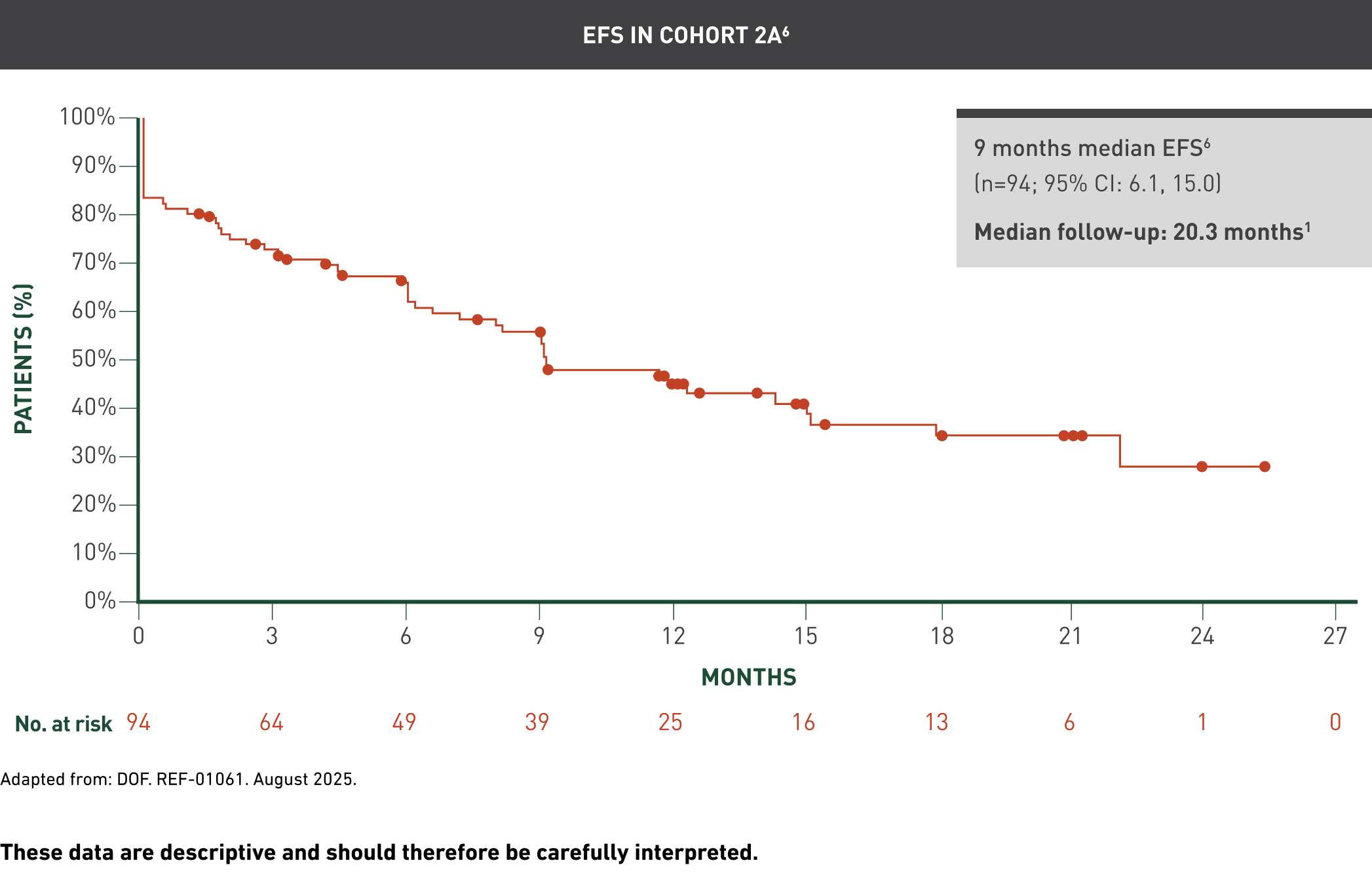
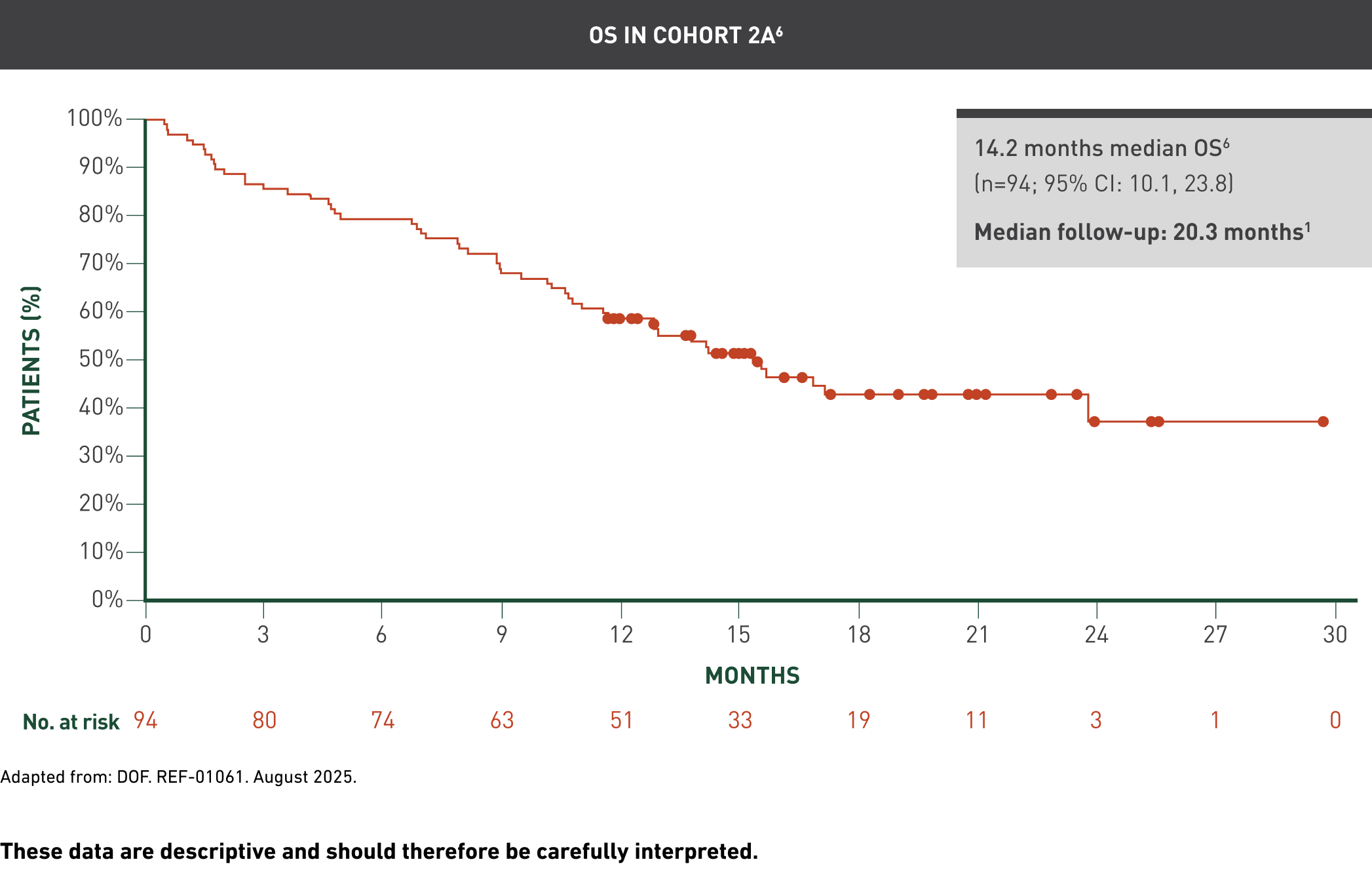
Cohort 2A has been specifically highlighted because it was the pivotal efficacy cohort in which the primary and key secondary endpoints were formally tested, while analysis of the total infused population provided supportive context across all treated patients.1
Overall remission in the total infused population1
Data cut-off: 7 February 2024; median follow-up: 32.8 months1
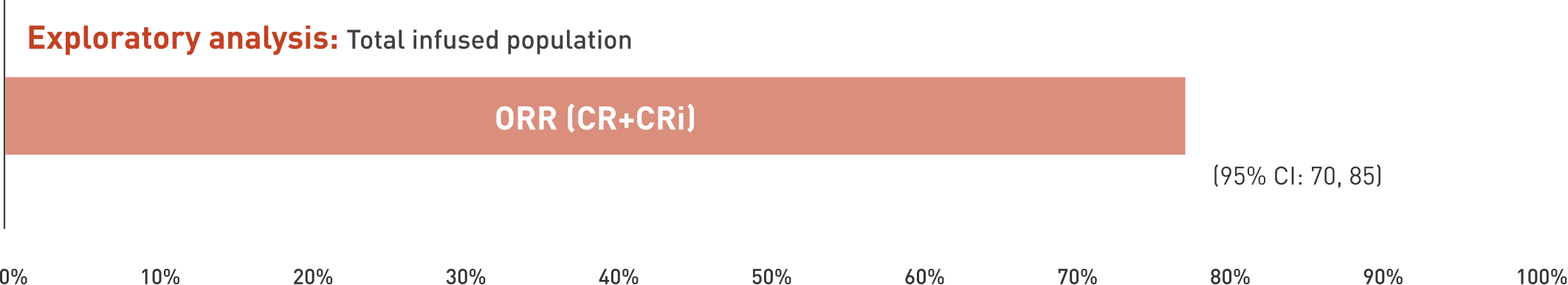
CR at any time1
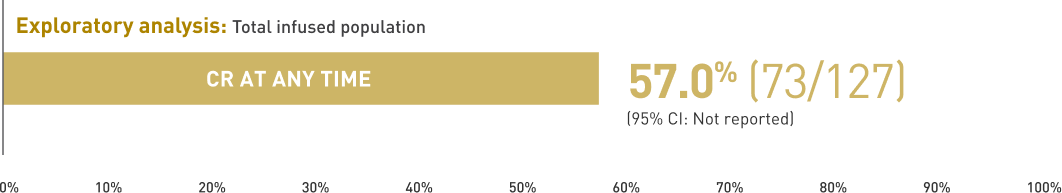
Selected secondary endpoints
Click on the buttons below to review other selected secondary endpoint data in the total infused population:
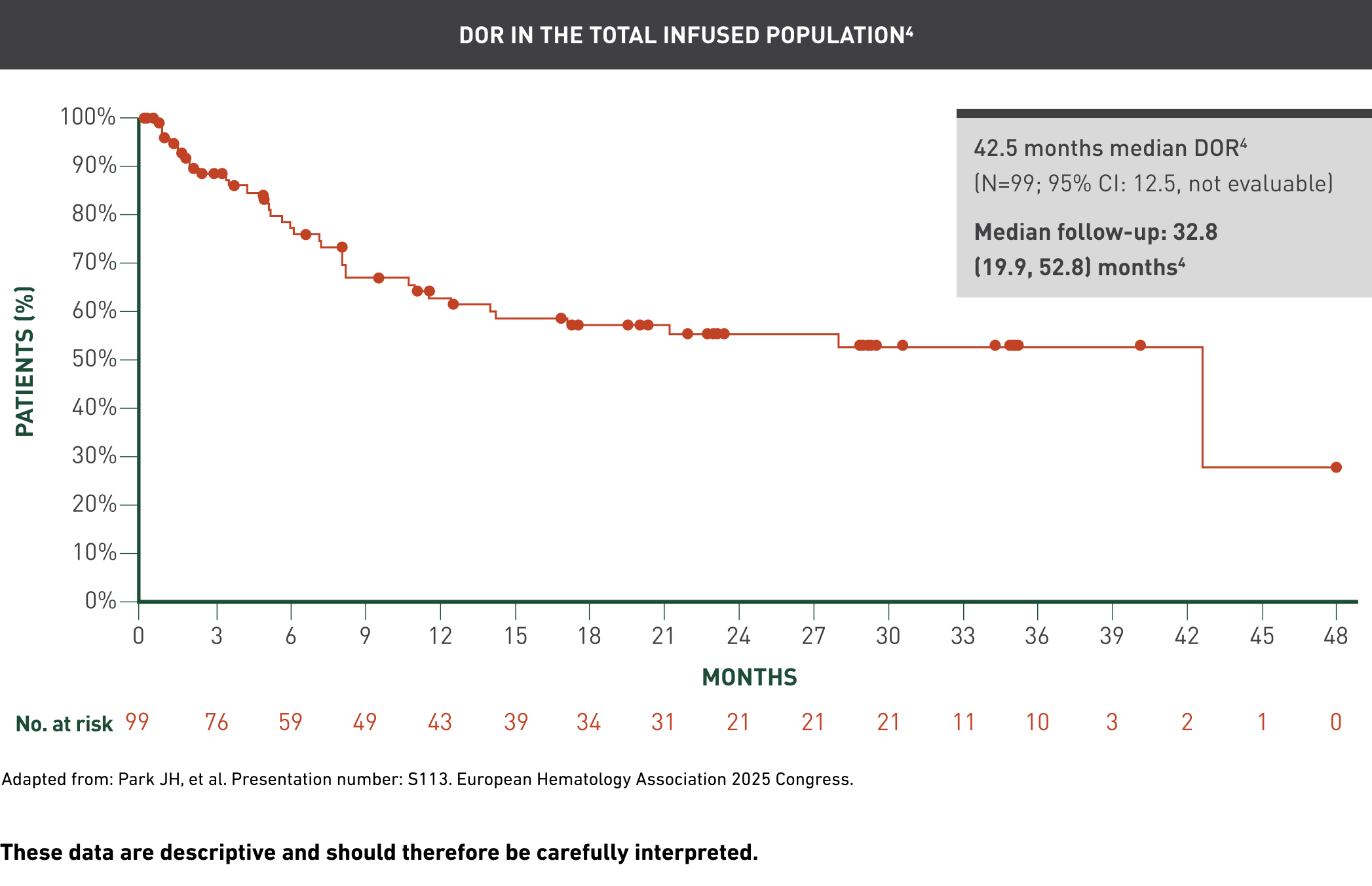
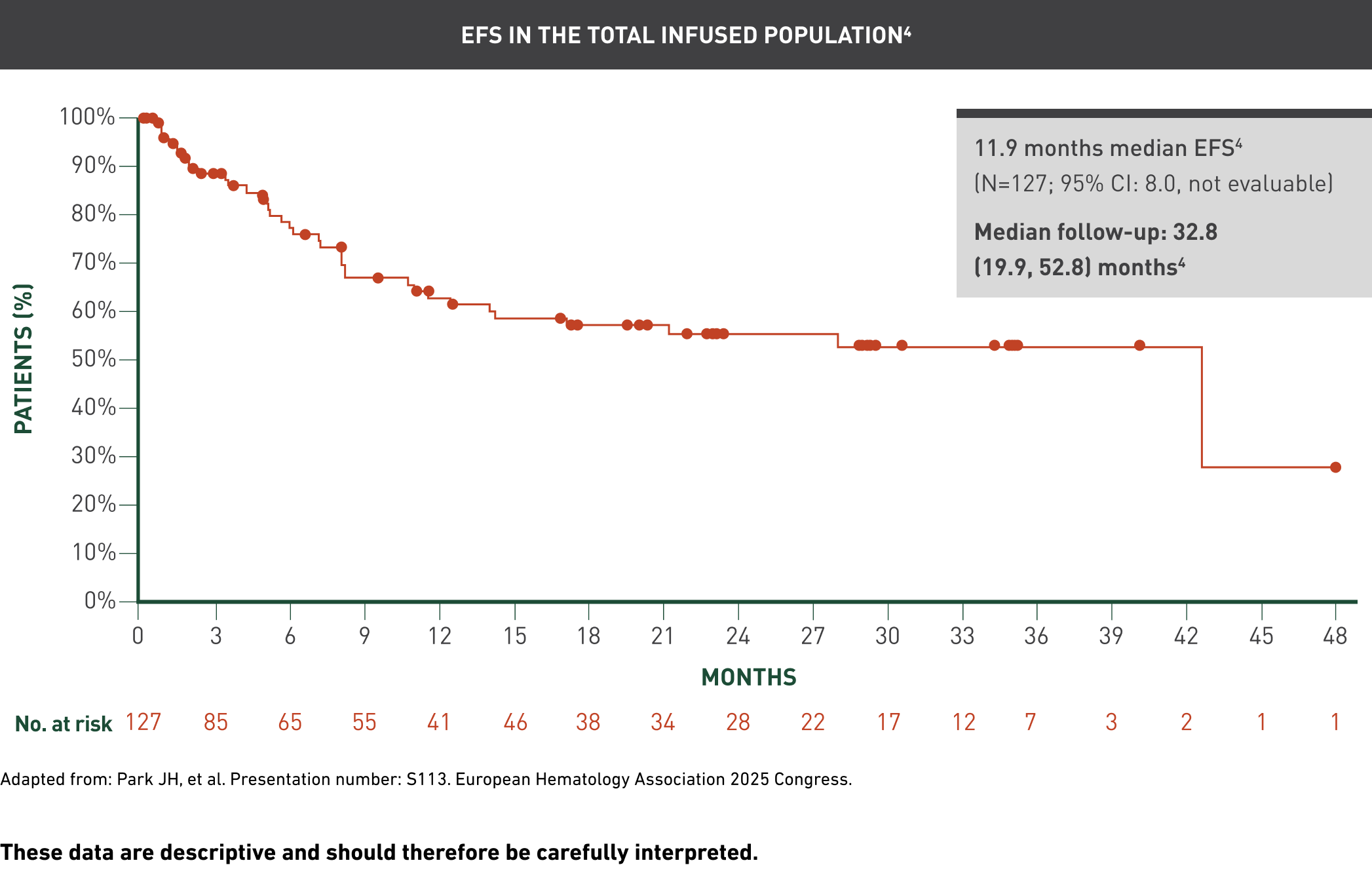
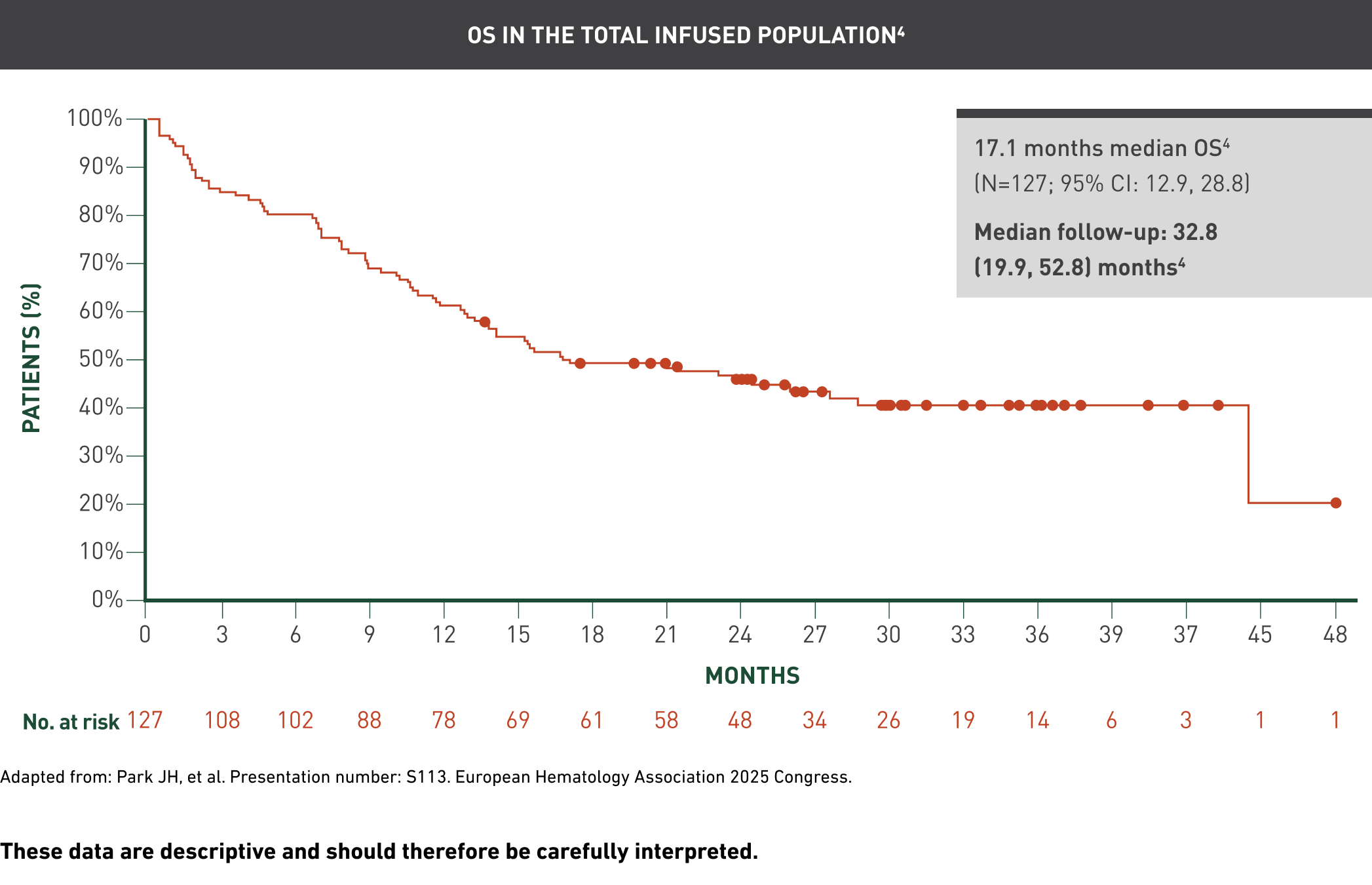
Overall remission rate1

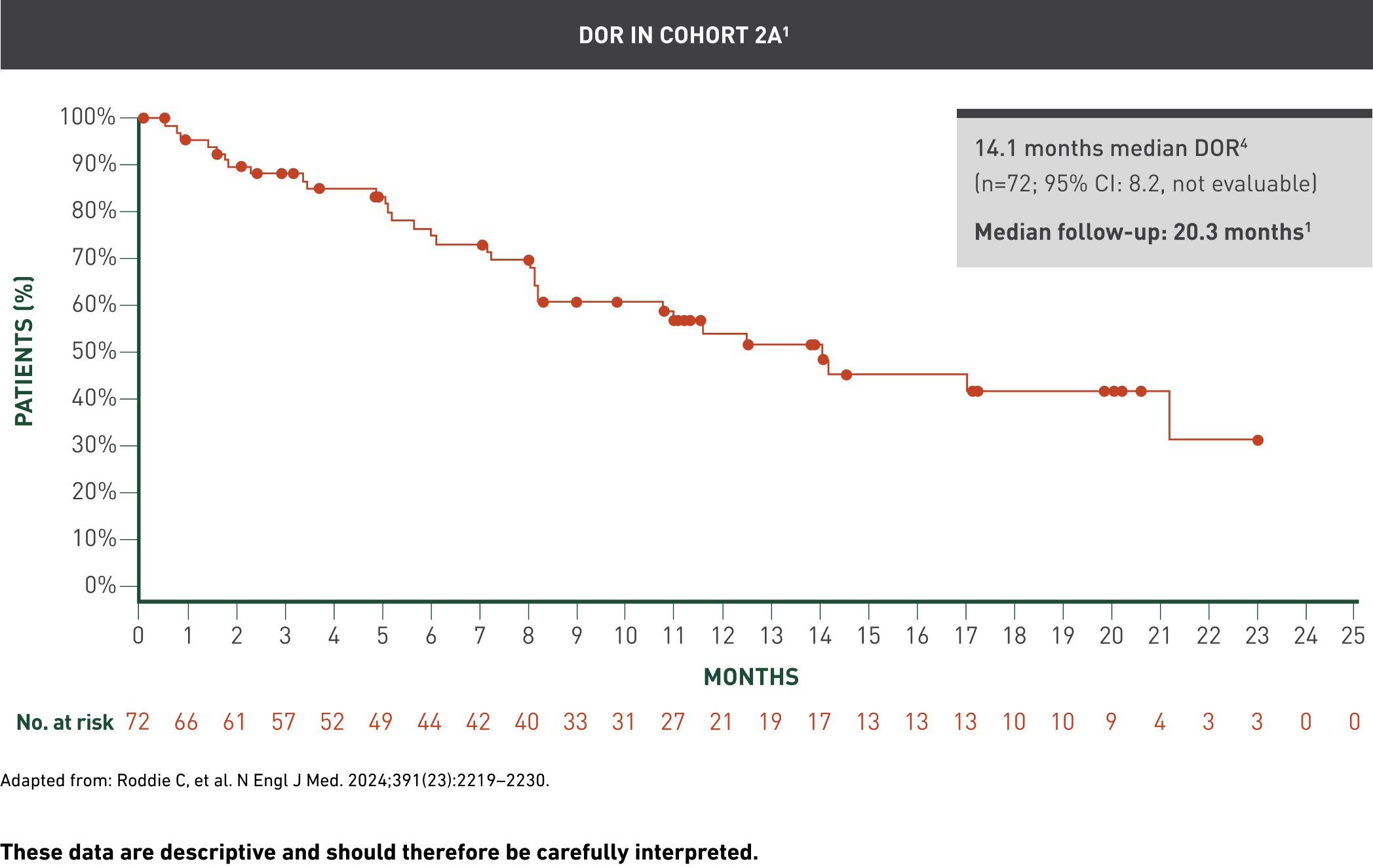
These data are descriptive and should therefore be carefully interpreted.
The median follow-up from the first AUCATZYL infusion to the data cutoff date in all the patients who received at least one infusion was 21.5 (range: 8.6, 41.4) months.1
CAR T persistency1,2

Maximum observed persistency1,2,5
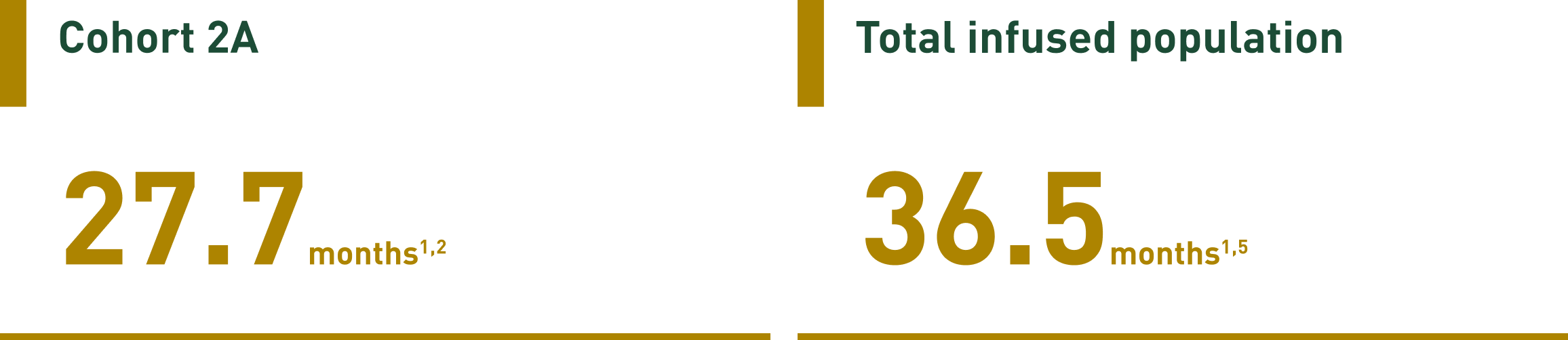
These data are descriptive and should therefore be carefully interpreted.
Severe* CRS and ICANS were observed in a small proportion of patients1,4
No new safety signals were observed at the ~3-year follow-up compared with the previous data cut:1,4
Median follow-up: | Median follow-up: | |||
|---|---|---|---|---|
Grade ≥3 | Any grade | Grade ≥3 | Any grade | |
CRS† | 2% (n=3/127) | 69% (n=87/127) | No change | No change |
ICANS† | 7% (n=9/127) | 23% (n=29/127) | No change | No change |
Infections | 52% (n=66/127) | 78% (n=99/127) | +4 (3%) | +4 (3%) |
Malignancies | 2% (n=2/127) | 2% (n=3/127) | No change | +2 (2%) |
New infections: Viral pneumonia (n=1), recurrent chest infection (n=1), enterocolitis infectious and hospital-acquired pneumonia (n=1), and multi-lobar pneumonia (n=1).4
New malignancies: Lentigo maligna melanoma (n=1) and urothelial transitional cell carcinoma (n=1); both were unrelated to AUCATZYL.4
Median time to onset after infusion (any grade) | Median duration | |
|---|---|---|
CRS† | 8 days | 5 days |
ICANS† | 12 days | 8 days |
After 11.3 additional months of follow-up, there have been no further incidences of CRS or ICANS reported.4
Treatment centres must have 24-hour immediate access to tocilizumab and emergency equipment must be available prior to infusion.
*Severe CRS and ICANS are defined as Grade ≥3.1
†Grading is based on the CTCAE v5.0.2
Summary of adverse reactions with AUCATZYL (N=127)2
In this summary, the median follow-up after being administered AUCATZYL is 21.5 (8.6, 41.4) months
The most common adverse reaction of any grade included CRS (69%), infections-pathogen unspecified (45%) and musculoskeletal pain (31%)
The most common non-laboratory Grade ≥3 adverse reactions were infections-pathogen unspecified (32%), febrile neutropenia (24%) and bacterial infectious disorders (11%)
The most common serious adverse reactions of any grade included infections-pathogen unspecified (28%), febrile neutropenia (13%) and ICANS (9%)
System order class | Frequency | Adverse reaction |
|---|---|---|
Infections and infestations | Very Common (≥1/10) | Infections-pathogen unspecified |
Blood and lymphatic system disorders | Very Common (≥1/10) | Neutropaenia* |
Immune system disorders | Very Common (≥1/10) | CRS |
Common (≥1/100 to <1/10) | Hypogammaglobulinaemia | |
Metabolism and nutrition disorders | Very Common (≥1/10) | Decreased appetite |
Psychiatric disorders | Common (≥1/100 to <1/10) | Delirium |
Nervous system disorders | Very Common (≥1/10) | Headache |
Common (≥1/100 to <1/10) | Tremor | |
Cardiac disorders | Very Common (≥1/10) | Tachycardia |
Common (≥1/100 to <1/10) | Arrhythmia | |
Vascular disorders | Very Common (≥1/10) | Hypotension |
Respiratory, thoracic and mediastinal disorders | Very Common (≥1/10) | Cough |
Gastrointestinal disorders | Very Common (≥1/10) | Nausea |
Common (≥1/100 to <1/10) | Stomatitis | |
Skin and subcutaneous tissue disorders | Very Common (≥1/10) | Rash |
Musculoskeletal and connective tissue disorders | Very Common (≥1/10) | Musculoskeletal pain |
General disorders and administration site conditions | Very Common (≥1/10) | Pyrexia |
Common (≥1/100 to <1/10) | Chills | |
Investigations | Very Common (≥1/10) | Alanine aminotransferase increased* |
Injury, poisoning and procedural complications | Common (≥1/100 to <1/10) | Infusion-related reaction |
Please consult the SmPC for full information.
*Frequency based on Grade 3 or higher laboratory parameter.2
AUCATZYL is contraindicated in patients with hypersensitivity to the active substance or to any of the excipients:
Disodium edetate
Dimethyl sulfoxide
Human albumin solution
Phosphate buffered saline:
Potassium chloride
Potassium dihydrogen phosphate
Sodium chloride
Disodium phosphate
Water for injections
Contraindications of the lymphodepleting chemotherapy must be considered.
Refer to the UK Public Assessment Report on the MHRA website for details of leukapheresis, lymphodepletion and bridging therapies used in the FELIX clinical trial for AUCATZYL.
Traceability
The traceability requirements of cell-based advanced therapy medicinal products must apply. To ensure traceability the name of the product, the batch number and the name of the treated patient must be kept for a period of 30 years after the expiry date of the product.
Autologous use
AUCATZYL is intended solely for autologous use and must not, under any circumstances, be administered to other patients. AUCATZYL must not be administered if the information on the product labels and Release for Infusion Certificate do not match the patient’s identity.
Monitoring
Patients must be monitored daily for 14 days after the first infusion for signs and symptoms of cytokine release syndrome, immune effector cell-associated neurotoxicity syndrome and other toxicities.
Frequency of monitoring after the first 14 days may be carried out at the physician’s discretion and continued for at least 4 weeks after the first infusion.
Patients must be instructed to remain within proximity of the qualified treatment centre for at least 4 weeks following the first infusion.
Reasons to delay treatment
Delay AUCATZYL treatment if there are unresolved serious adverse reactions from preceding chemotherapies, if the patient is experiencing severe intercurrent infection, or has active graft-versus-host disease. If the patient requires supplementary oxygen, AUCATZYL should only be infused, if considered appropriate, based on the treating physician’s benefit/risk assessment.
Reasons to delay the second split dose
Dosage delays or discontinuation may be required after the first split dose to manage adverse reactions.
Patients with Grade 2 cytokine release syndrome and/or Grade 1 immune effector cell-associated neurotoxicity syndrome following the first split dose may receive the second dose on Day 10 (± 2 days) up to Day 21 only if cytokine release syndrome has resolved to Grade 1 or less and immune effector cell-associated neurotoxicity syndrome has completely resolved.
For patients with Grade ≥3 (i) severe infection at the time of infusion of AUCATZYL or (ii) requirement for supplementary oxygen or (iii) other clinically relevant adverse reactions following the first split dose: consider postponing AUCATZYL up to Day 21 to allow the situation to resolve.
In addition, the second split dose is not to be administered if ≥Grade 3 cytokine release syndrome, ≥Grade 2 immune effector cell-associated neurotoxicity syndrome and/or ≥Grade 3 pulmonary or cardiac toxicities are observed following the first split dose.
Grading is based on the Common Terminology Criteria for Adverse Events (CTCAE) v5.0.
Cytokine release syndrome
Refer to local institutional/national guidelines for advice on monitoring and management of cytokine release syndrome.
Evaluation for haemophagocytic lymphohistiocytosis/macrophage activation syndrome is to be considered in patients with severe or unresponsive cytokine release syndrome. Treatment should be administered per institutional standards.
Availability of tocilizumab
Treatment centres must have 24-hour immediate access to tocilizumab and emergency equipment must be available prior to infusion. In the exceptional case where tocilizumab is not available owing to a shortage, then alternatives to tocilizumab to treat cytokine release syndrome must be available prior to infusion. Shortages of tocilizumab may be checked for in the MHRA Central Alerting System.
Immune effector cell-associated neurotoxicity syndrome
Patients should be monitored for signs and symptoms of immune effector cell-associated neurotoxicity syndrome.
Refer to local institutional/national guidelines for advice on monitoring and management of immune effector cell-associated neurotoxicity syndrome.
Prolonged cytopenias
Patients may exhibit cytopenias for several weeks following lymphodepleting chemotherapy and AUCATZYL infusion and should be managed according to institutional guidelines.
Patient blood counts must be monitored after AUCATZYL infusion.
Severe Infections
AUCATZYL should not be administered to patients with clinically significant active systemic infections.
Severe infections, including life-threatening or fatal infections occurred in patients after receiving AUCATZYL.
Grade 3 or higher febrile neutropenia was observed in patients after AUCATZYL infusion and may be concurrent with cytokine release syndrome.
Patients with human immunodeficiency virus (HIV), hepatitis B virus (HBV) and hepatitis C virus (HCV) infection
There is no clinical experience in patients with a positive test for HIV, active HBV, or active HCV infection. Screening for HBV, HCV, HIV and other infectious agents must be performed in accordance with clinical guidelines before collection of cells for manufacturing.
Leukapheresis material from patients with active HIV, active HBV, or active HCV infection will not be accepted for manufacturing.
Viral reactivation
Viral reactivation, e.g., HBV reactivation, can occur in patients treated with medicinal products directed against B cells and could result in fulminant hepatitis, hepatic failure, and death.
Hypogammaglobulinaemia
Hypogammaglobulinaemia is caused by B cell aplasia and has been seen as a consequence of depletion of normal B cells by CAR T cell therapy. Hypogammaglobulinaemia can occur in patients treated with AUCATZYL.
Hypogammaglobulinaemia predisposes patients to become more susceptible to infections. Immunoglobulin levels should be monitored after treatment with AUCATZYL and managed per institutional guidelines including infection precautions, antibiotics or antiviral prophylaxis and immunoglobulin replacement.
Prior stem cell transplantation (graft versus host disease)
It is recommended that patients do not receive AUCATZYL within 3 months of undergoing an allogeneic stem cell transplantation because of the potential risk of AUCATZYL worsening graft versus host disease. There must be a gap of 3 months after allogeneic stem cell transplantation before leukapheresis is carried out to obtain material to manufacture AUCATZYL.
Stem cell transplantation after CAR T cell therapy
The role of allogeneic stem cell transplant following CAR T-cell therapy is unclear. Note: a chemotherapy-based preparative regimen associated with a subsequent stem cell transplant procedure will neutralise the effect of CAR T-cells.
Secondary malignancies including of T-cell origin
Patients treated with AUCATZYL may develop secondary malignancies. T-cell malignancies have been reported following treatment of haematological malignancies with a BCMA- or CD19-directed CAR T-cell therapy. T-cell malignancies, including CAR-positive malignancies, have been reported within weeks and up to several years following administration of a CD19- or BCMA-directed CAR T-cell therapy. There have been fatal outcomes. Patients should be monitored life-long for signs of secondary malignancies. In the event that a secondary malignancy occurs, the company should be contacted to obtain instructions on the collection of patient samples for testing.
Tumour lysis syndrome
Tumour lysis syndrome, which may be severe, has occasionally been observed in the FELIX trial and with other CAR T-cell products. To minimise the risk of tumour lysis syndrome, patients with high tumour burden should receive tumour lysis syndrome prophylaxis as per standard guidelines prior to AUCATZYL infusion. Signs and symptoms of tumour lysis syndrome after AUCATZYL infusions must be monitored, and events managed according to standard guidelines.
Hypersensitivity reactions
Serious hypersensitivity reactions, including anaphylaxis, may occur due to dimethyl sulfoxide in AUCATZYL.
Transmission of an infectious agent
Although AUCATZYL is tested for Sterility and Mycoplasma, a risk of transmission of infectious agents exists. Healthcare professionals administering AUCATZYL must, therefore, monitor patients for signs and symptoms of infection after treatment and treat appropriately, if needed.
Interference with virological testing
Due to limited and short spans of identical genetic information between the lentiviral vector used to create AUCATZYL and HIV, some HIV nucleic acid tests may give a false positive result.
Blood, organ, tissue and cell donation
Patients treated with AUCATZYL must not donate blood, organs, tissues and cells for transplantation.
Patient Card
The Patient Card must be given to the patient after treatment.
Sodium content
This medicinal product contains 1131 mg sodium per target dose, equivalent to 57% of the WHO recommended maximum daily intake of 2 g sodium for an adult.
Potassium content
This medicinal product contains 39 mg potassium per target dose, equivalent to 1% of the WHO recommended maximum daily intake of 3.51 g potassium for an adult.
Long-term follow-up
Patients are expected to be enrolled in a long-term follow-up scheme in order to better understand the long-term effects of AUCATZYL.
Paediatric population
There is not any clinical experience of AUCATZYL in paediatric patients. No specific guidance for use in this patient population exists.
No interaction studies have been performed.
Some patients required tocilizumab and/or corticosteroids for the management of cytokine release syndrome and immune effector cell-associated neurotoxicity syndrome.
Prophylactic use of systemic corticosteroids may interfere with the activity of AUCATZYL. Prophylactic use of systemic corticosteroids is therefore not recommended before infusion.
Patients with high tumour burden (≥20%) had a greater frequency of cytokine release syndrome, which was managed by the use of tocilizumab and/or corticosteroids. Patients with a higher tumour burden showed a more robust CAR T-cell expansion, which is known to increase the likelihood of occurrence of cytokine release syndrome or immune effector cell-associated neurotoxicity syndrome. Administration of tocilizumab or corticosteroids for the treatment of cytokine release syndrome and immune effector cell-associated neurotoxicity syndrome did not affect the rate or extent of expansion and persistency.
Live vaccines
The safety of immunisation with live viral vaccines during or following treatment with AUCATZYL has not been studied. As a precautionary measure, vaccination with live vaccines is not recommended for at least 6 weeks prior to the start of lymphodepletion chemotherapy, during AUCATZYL treatment, and until immune recovery following treatment. Refer to local institutional/national guidance for advice on live vaccines.
Bridging therapies
Blinatumomab was not permitted as a bridging therapy in the FELIX clinical study, and there is no clinical experience with use of this product as a bridging therapy before AUCATZYL treatment.
Herbal remedies with immunomodulatory properties
There are no formal interaction studies with herbal remedies and AUCATZYL; general precautions are recommended due to their potential immunomodulatory effects.
Women of childbearing potential/Contraception in males and females
The pregnancy status of women of childbearing potential must be verified before starting AUCATZYL treatment.
See the prescribing information for fludarabine and cyclophosphamide for information on the need for effective contraception in patients who receive the lymphodepleting chemotherapy.
There is insufficient exposure data to provide a recommendation concerning duration of contraception following treatment with AUCATZYL.
For females who are not postmenopausal (<24 months of amenorrhea) or who are not surgically sterile (absence of ovaries and/or uterus), two methods of contraception, comprising of one highly effective method of contraception together with a barrier method, must be used during the treatment period and for at least 12 months after the last dose of AUCATZYL. They must agree not to donate eggs (ova, oocytes) for the purposes of assisted reproduction during treatment and for 12 months after receiving the last dose of AUCATZYL.
Pregnancy
There are limited data available with the use of AUCATZYL in pregnant women. No animal reproductive and developmental toxicity studies have been conducted with AUCATZYL to assess whether it can cause foetal harm when administered to a pregnant woman.
It is not known if AUCATZYL has the potential to be transferred to the foetus. Based on the mechanism of action, if the transduced cells cross the placenta, they may cause foetal toxicity, including B-cell lymphocytopenia. Therefore, AUCATZYL is not recommended for women who are pregnant, or for women of childbearing potential not using contraception. Pregnant women must be advised on the potential risks to the foetus.
Pregnancy after AUCATZYL therapy must be discussed with the treating physician.
Assessment of immunoglobulin levels and B-cells in newborn infants of mothers treated with AUCATZYL must be considered.
Breastfeeding
It is unknown whether AUCATZYL cells are excreted in human milk or transferred to the breastfeeding child. Breastfeeding women must be advised of the potential risk to the breast-fed child.
Fertility
There are very limited data on the effect of AUCATZYL on fertility. Effects on male and female fertility have not been evaluated in animal studies.
AUCATZYL may have a major influence on the ability to drive and use machines.
Because of the potential for neurological events, including altered mental status or seizures, patients must refrain from driving or operating heavy or potentially dangerous machines until at least 8 weeks after infusion or until resolution of the neurological event as confirmed by the treating physician.


Process and administration
Learn more about tumour-guided split dosing and how to administer AUCATZYL.
Stay updated
Sign up to receive the latest news and product updates from Autolus.
By clicking ‘Receive key updates’ you’re confirming that you agree with our Terms and conditions.
Aucatzyl is indicated for the treatment of adult patients (≥18 years) with relapsed or refractory B-cell precursor acute lymphoblastic leukaemia.2
NICE recommends AUCATZYL to treat relapsed or refractory B-cell precursor acute lymphoblastic leukaemia in people aged ≥26 years in England, Wales and Northern Ireland.7,8
ALL=acute lymphoblastic leukaemia; allo-SCT=allogeneic stem cell transplant, B-ALL=B-cell precursor acute lymphoblastic leukaemia, BCMA=B-cell maturation antigen, BM=bone marrow, CAR=chimeric antigen receptor, CI=confidence interval, CNS=central nervous system, CR=complete remission, CRi=complete remission with incomplete haematologic recovery, CRS=cytokine release syndrome, CTCAE=Common Terminology Criteria for Adverse Events, DOR=duration of response, EFS=event-free survival, EMD=extramedullary disease, HBV=hepatitis B, HCV=hepatitis C, HIV=human immunodeficiency virus, ICANS=immune effector cell-associated neurotoxicity syndrome, MHRA=Medicines and Healthcare products Regulatory Agency, MRD=minimal residual disease, NICE=National Institute for Health and Care Excellence, ORR=overall remission rate, OS=overall survival, R/R=relapsed/refractory; WHO=World Health Organization.
Roddie C, et al. Obecabtagene autoleucel in adults with B-cell acute lymphoblastic leukemia. N Engl J Med. 2024;391(23):2219–2230.
Summary of Product Characteristics, Aucatzyl 2025.
Summary of Product Characteristics, Tecartus 2025.
Park JH, et al. Can CAR T-cell therapy be a definitive treatment for adult R/R B-ALL without transplant? Long-term findings and predictors of sustained remission for obecabtagene autoleucel. Presentation number: S113. European Hematology Association 2025 Congress. 12–15 June, 2025. Milan, Italy.
Roddie C, et al. Obecabtagene autoleucel in adults with B-cell acute lymphoblastic leukemia. N Engl J Med. 2024;391(23):2219–2230. Supplementary Appendix.
Autolus. DOF. REF-01061. August 2025.
NICE. Obecabtagene autoleucel for treating relapsed or refractory B-cell precursor acute lymphoblastic leukaemia. Issued December 2025. Accessed March 2026. https://www.nice.org.uk/guidance/ta1116/resources/obecabtagene-autoleucel-for-treating-relapsed-or-refractory-bcell-precursor-acute-lymphoblastic-leukaemia-pdf-2973530453903557.
AWTTC. Obecabtagene autoleucel (Aucatzyl®). Issued November 2025. Accessed March 2026. https://awttc.nhs.wales/accessing-medicines/medicine-recommendations/obecabtagene-autoleucel-aucatzyl/.


